Tomai lab. (FRIS)


The high controllability of solvent properties is
the advantage of supercritical fluid. For example,
supercritical water is miscible with organic compounds.
The unique characteristics of supercritical fluids enable
the synthesis of unique nanomaterials that cannot be
synthesized under conventional synthesis process. Our
research group aims to develop innovative chemical
process driven by low-temperature waste heat and
renewable energy by designing unique nanomaterial.
We are working on improving process/device by
precisely controlling multi-scale hierarchical structure
of nanomaterial from the composition and crystalline
structure to the dispersion/aggregation (porous)
structure.
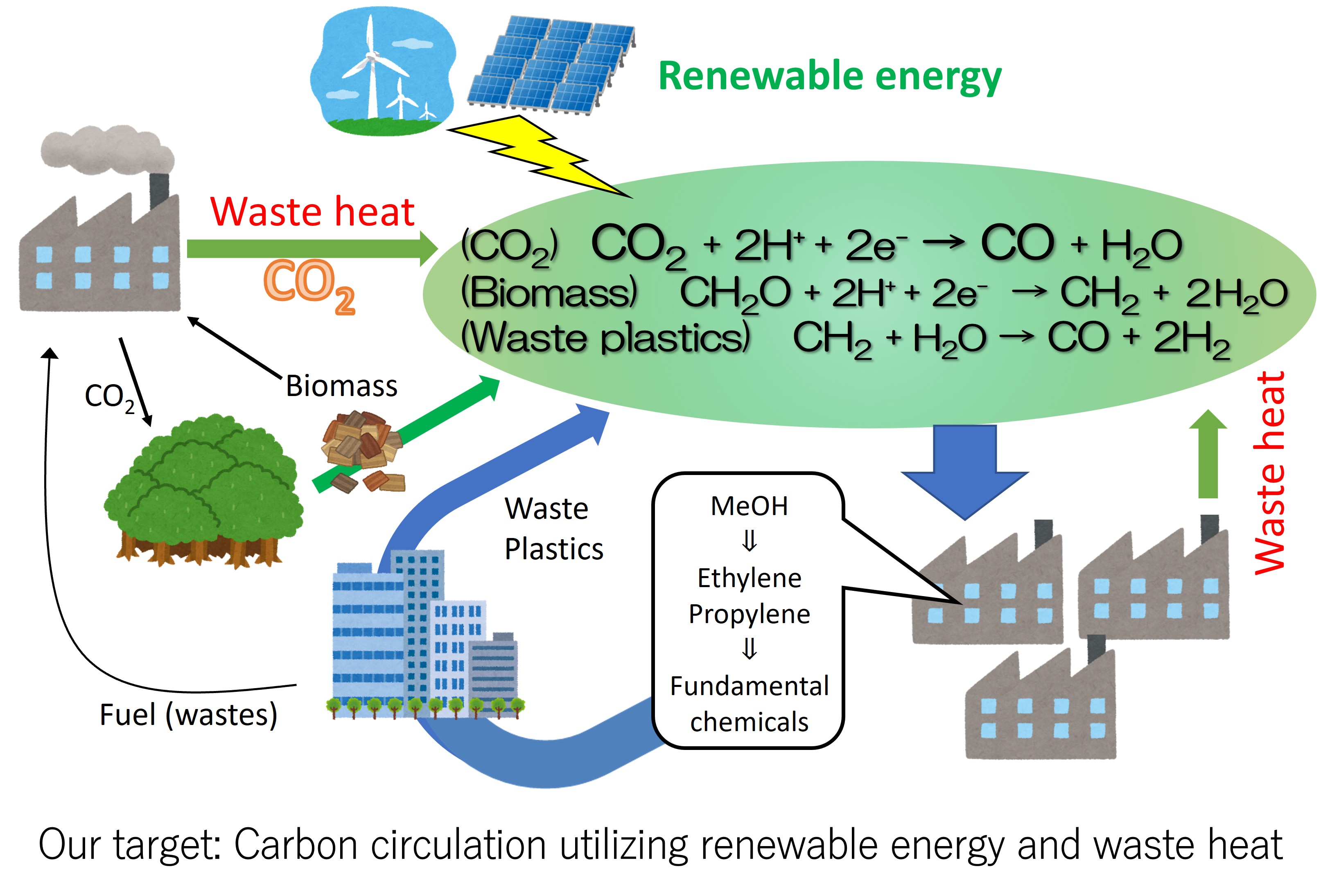
Moreover, we are developing electrochemical materialenergy
conversion systems using supercritical and
subcritical fluids as reaction fields by focusing on their
unique properties, specifically carbon dioxide and water
electrolysis. Through our research efforts, we aim to
establish scientific principles for high-temperature, highpressure
electrochemical energy conversion systems
and contribute to realizing a sustainable society.